 My message to others is, don’t lose hope,” the survivor said. “I couldn’t believe I was alive when I first opened my eyes. Dr Trivedi said he had been started on physiotherapy. Dr Karnik said an MRI revealed that a part of the brain had developed lesions. The cyanide antidote kit consists of three medications given together: amyl nitrite, sodium nitrite, and sodium thiosulfate. 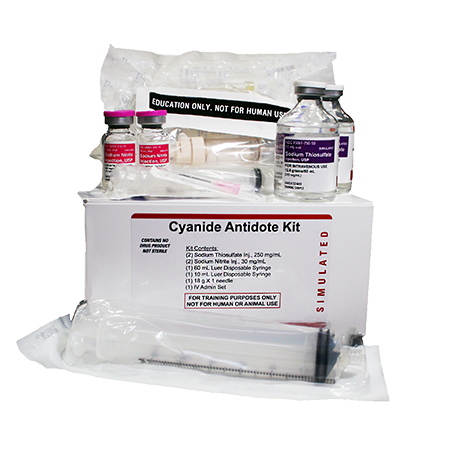
The student has developed tremors in his hands and slurred speech. “My son was born in Sion hospital, so they have given him a second lease of life,” the student’s mother said. He came off the ventilator on November 4 and was moved out of the ICU by November 7. His health started turning around after 48 hours. Dr Karnik said as the 23-year-old student was unconscious, they had to give him the amyl nitrite through the ventilator tubes. Indication-specific dosing for cyanide antidote kit (sodium thiosulfate, sodium nitrite, & amyl nitrite), frequency-based adverse effects, comprehensive interactions, contraindications, pregnancy & lactation schedules, and cost information. These kits have a shelf life of only a year. After the death, the institute head had donated an antidote kit to Sion hospital. Dr Karnik recalled treating the last case of cyanide poisoning a decade ago, an engineering student from a premier institute. Fortunately, he had told his friend about taking cyanide” Dr Trupti Tridevi, in-charge of the medicine ICU at LTMG, said. 
We could give him hydroxocobalamin, a form of vitamin B12, and that helped too. Argote-Araméndiz, Alejandra Caycedo, in Ciottones Disaster Medicine (Third Edition), 2024 Cyanide Antidote Kits (CAK) Cyanide antidote kits were commercially available kits that included three different components: amyl nitrite, sodium nitrite, and sodium thiosulfate. We gave medicines to support his blood pressure and heart functions. The key here was to keep him oxygenated, ventilated and hydrated till the antidote came. “Cyanide starts to deprive tissues of oxygen. An online search revealed that a Hyderabad chemist had it. The agency has not received any complaints of harmful reactions from the drugs.After a night-long search, the doctors and the family realised that the city did not have a single unit of the cyanide antidote kit that comprises amyl nitrite, sodium nitrite, and sodium thiosulfate. 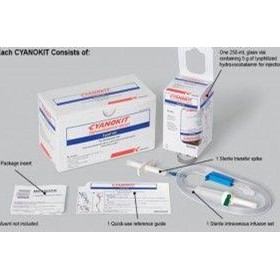
The FDA said hospitals that bought the kits should return them to Keystone. The company’s Web site advertises the kits but does not list the price. Keystone officials did not return a call Thursday. “This action is a significant step in protecting the public health.” 3004909000, 1 KIT SODIUM CYANIDE ANTIDOTE KIT CONSIST OF THE FOLLOWING: 6 X 0.3ML - AMYL NITRATE INHALATION 2 X 10ML - SODIUM NITRATE INSTRVENOUS INJECTION. 
“The FDA is taking this action because Keystone has refused to take these unapproved products off the market,” said Michael Chappell, the FDA’s acting associate commissioner for regulatory affairs. Treatment typically involves injection of a form of vitamin B-12 that helps the body eliminate the cyanide through urination. The kits were sold to hospitals although the drugs, made by a San Diego company contracted by Keystone, have not been proven “safe and effective,” the FDA said.Ĭyanide poisoning most commonly occurs after inhaling smoke in a residential fire or through fumes during industrial accidents. Keystone Pharmaceuticals refused to heed two warnings from the FDA to stop selling kits for the treatment of cyanide poisoning, according to federal officials. Federal marshals seized $39,000 worth of unapproved drugs Thursday from a Laguna Hills company, the Food and Drug Administration said. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
